|
5/16/2023 0 Comments Bohr atomic model
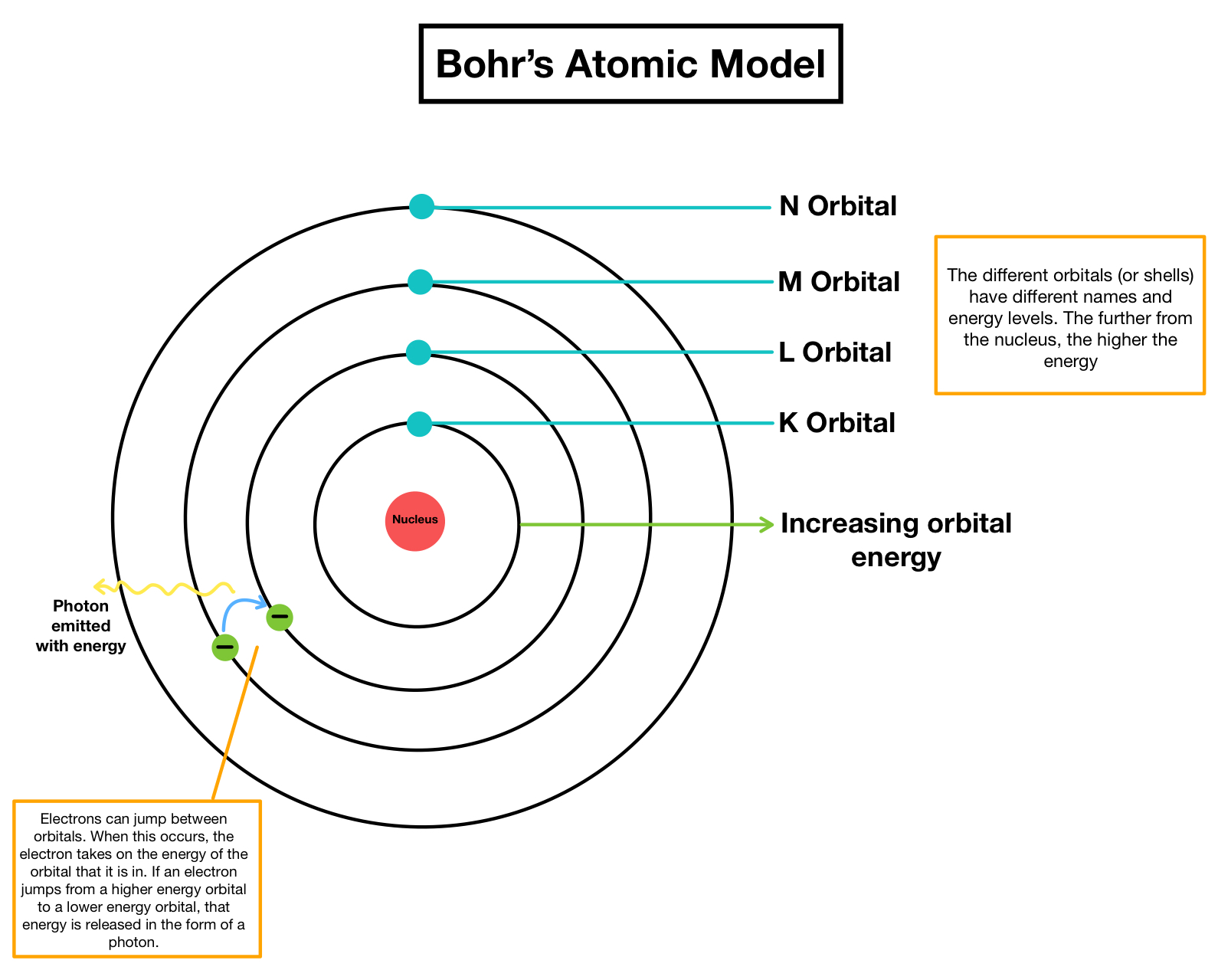
It failed to make correct predictions of large-sized atoms and provided sufficient information only for smaller atoms.The Bohr model: Journey to find structure of atomsīritish physicist Joseph John Thomson made the first major breakthrough in the understanding of atoms in 1897 when he discovered that atoms contained tiny negatively charged particles that he called electrons. Bohr’s model could not explain shapes and geometry molecules.However, according to Bohr, electrons move in a well-defined circular orbit. According to this principle, it is impossible to determine simultaneously the exact position and momentum of a small moving particle like an electron with absolute certainty. It does not obey Heisenberg’s uncertainty principle.This is based on de Broglie’s suggestion that electrons of light have a dual character. But modern research reveals that an electron moves around the nucleus in three-dimensional space. According to Bohr, the circular orbits of electrons are planar.It failed to explain the Stark effect when the spectral lines get split into fine lines under the influence of an electric field.It could not explain the Zeeman effect when the spectral lines are split into closely spaced lines under the influence of magnetic field. 
It could not explain the atomic spectra of elements having more than one electron.The Limitations of Bohr’s atomic model are as follows: 
Read Also: Planck Quantum Theory and Black Body Radiation
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
